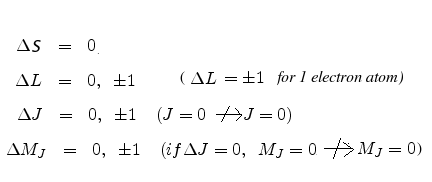In normal Zeeman effect, we studied the splitting of spectral lines into three components in an one electron atom in the presence of a weak magnetic field. Although not stated explicitly, we had then assumed erroneously that the electron did not have any spin. Actually, normal Zeeman Effect is observed in multi-electron atoms with even number of optically active electrons, the electronic transitions occuring between Singlet states (Total spin quantum number S=0, multiplicity=2S+1=1), as for example between 1P and 1S states.
On the other hand when electronic transitions occur in single or multi-electron atoms in the presence of weak magnetic fields between Multiplet states (Total spin quantum number S=1/2, multiplicity=2S+1=2 (Doublet), S=1, multiplicity=2S+1=3 (Triplet) etc), as for example between 2P3/2 and 2S1/2 states, we observe many more lines and the effect is called anomalous Zeeman effect. It can be explained through the LS coupling of the individual angular momentum vectors and the behaviour of the resultant total angular momentum vector and the total magnetic dipole moment in the presence of the external magnetic field.
The term 'anomalous' originates from the fact that the total magnetic moment
![]() =
= ![]() +
+ ![]() is not antiparallel (and therefore not collinear) to the total angular momentum
is not antiparallel (and therefore not collinear) to the total angular momentum ![]() . This is because of the gyromagnetic ratio having a value
. This is because of the gyromagnetic ratio having a value ![]() 1.
1.
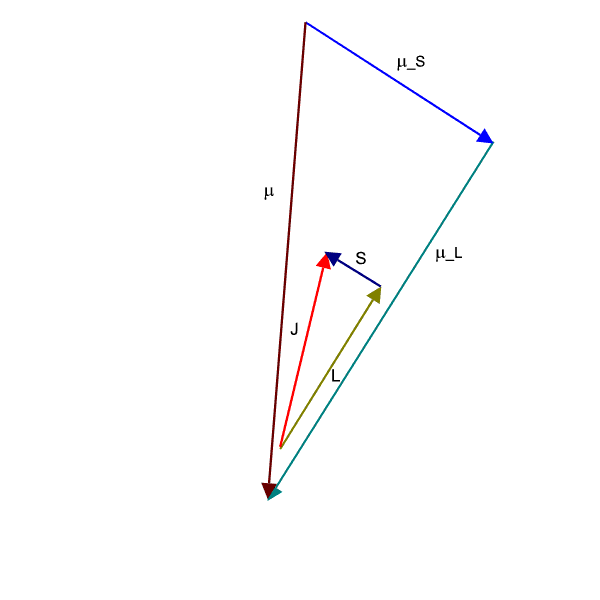
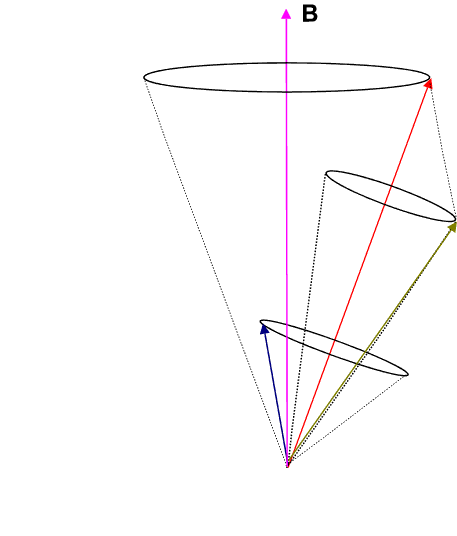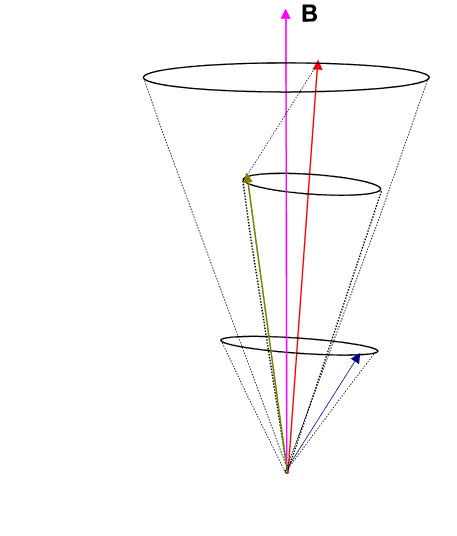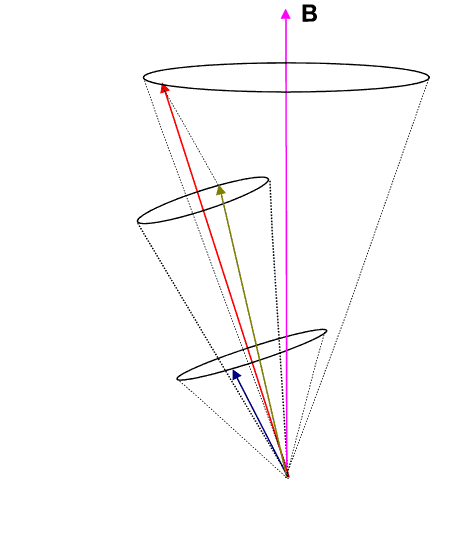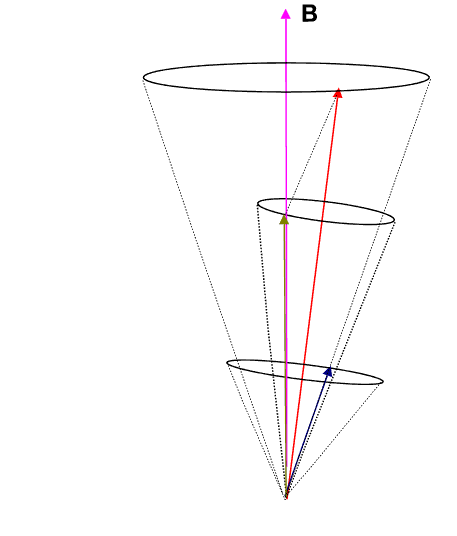
If we employ the vector model as has been done in the animation below, we observe that the ![]() (brownish-yellow) and
(brownish-yellow) and ![]() (deep-blue) vectors precess about
(deep-blue) vectors precess about ![]() (red) (due to spin-orbit coupling), which in turn precesses slowly about the external magnetic field
(red) (due to spin-orbit coupling), which in turn precesses slowly about the external magnetic field ![]() (pink) which lies parallel to the z-axis.
(pink) which lies parallel to the z-axis.
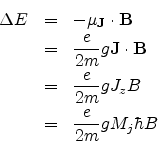
where g is called the Lande g-factor and is given by
![]() Thus each energy level splits into 2J + 1 levels, since MJ=-J to +J in integral steps.
Thus each energy level splits into 2J + 1 levels, since MJ=-J to +J in integral steps.
It is very important to note that for singlet states the spin quantum number S=0 so that J=L and the Lande g-factor becomes equal to the gyromagnetic ratio=2. Hence the above formulation reduces to that of the normal Zeeman effect which was explained earlier in terms of only the orbital angular momentum and its associated magnetic moment.
The emission spectrum of the atom placed in the external magnetic field ![]() will show additional lines apart from the original line which corresponds to the situation when
will show additional lines apart from the original line which corresponds to the situation when ![]() =0. The pertinent selection rules for electric dipole transitions are
=0. The pertinent selection rules for electric dipole transitions are